
In chromogenic detection, antibody-epitope binding is visualized using an enzyme conjugated to the secondary antibody or to the detection system (e.g. ABC method). The enzyme, either horseradish peroxidase (HRP) or alkaline phosphatase (AP), catalyzes the conversion of its respective chromogenic substrate to a colored precipitate at the site of antibody binding.
SYSY's reference protocols use HRP-based chromogenic detection with 3,3’-Diaminobenzidine (DAB) as the chromogenic substrate. DAB is extremely stable and stained slides are stable for years. Many cells (e.g. erythrocytes, granulocytes, neurons) have endogenous peroxidases that need to be blocked to reduce background staining. SYSY suppresses endogenous peroxidase with a 3% H2O2 blocking step before primary antibody incubation. However, some epitopes can be destroyed by high concentrations of hydrogen peroxidase leading to reduced or no antibody-antigen binding. In this case, the 3% H2O2 blocking step can be performed after primary antibody incubation.
No 3% H2O2 blocking step is required when using Alkaline Phosphatase (AP) or fluorescent detection.
DAB stained slides can be dehydrated, cleared, and mounted with an organic mounting medium as delineated in our SYSY's reference protocol. Sections mounted in organic mounting media often have better optical quality than sections mounted in aqueous mounting media.
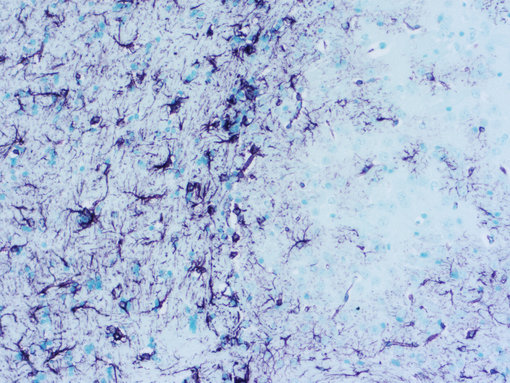
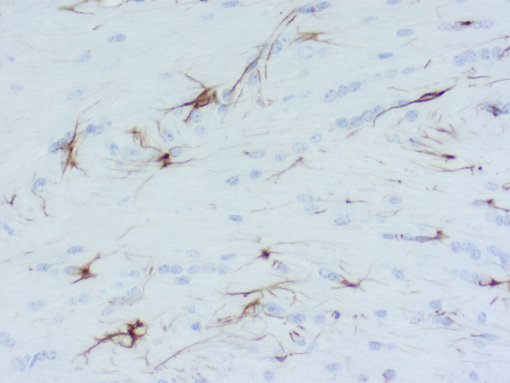
Figure 1: Chromogenic detection using ImmPACT VIP (purple) as HRP substrate. Counterstain: methylene green (light green).
Figure 2: Chromogenic detection using DAB (brown) as HRP substrate. Counterstain: hematoxylin (blue).
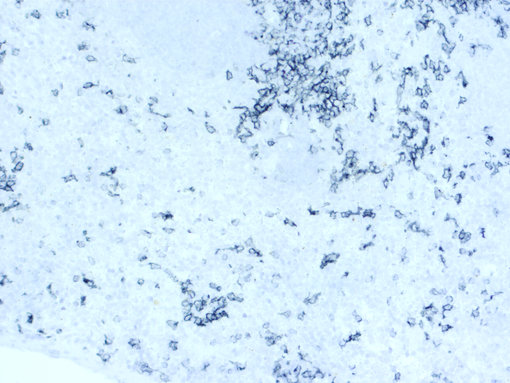
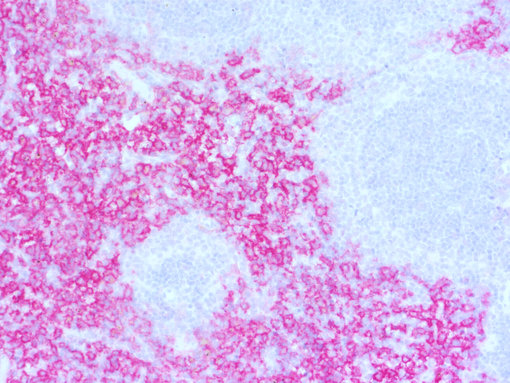
Figure 3: Chromogenic detection using ImmPACT SG (grey) as HRP substrate. Counterstain: hematoxylin (blue).
Figure 4: Chromogenic detection using ImmPACT AP RED (red) as AP substrate. Counterstain: hematoxylin (blue).
In Immunofluorescence, fluorophore-conjugated secondary antibodies are used to localize antigen-antibody complexes. Fluorescent detection allows easier Multiplexing, especially for co-localized targets, and has a higher dynamic range when high and low abundance targets have to be visualized on one slide. However, many FFPE tissues, especially spleen and kidney, show high tissue autofluorescence making the interpretation of assay results particularly difficult in green- and red-channel fluorophores.